Vaccine Effectiveness in the USA
With 80% of Americans vaccinated, it is clear that the vaccines are working. In fact, vaccinated people are much less likely to experience hospitalization and deaths as a result of COVID-19.
Latest data shows that unvaccinated people are 17x more likely than vaccinated people to be hospitalized with COVID-19. And they are as much as 15x more likely to die from the disease. Of the people who died from COVID during the past month in the USA, 99% of them were unvaccinated.

COVID vaccines are also effective against new coronavirus variants, including Delta, Lambda, and Mu, even though these new variants are much more contagious than the previous strain.
Though vaccinating does not guarantee you will not get COVID, it will help you not get as sick. This is especially true for children 12+ who are being affected by the new variants much more than the original strain.
Vaccine Development
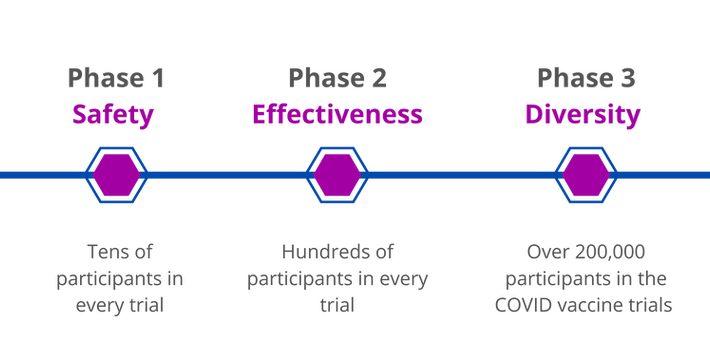
COVID-19 vaccines are the most thoroughly tested vaccines in human history for any disease.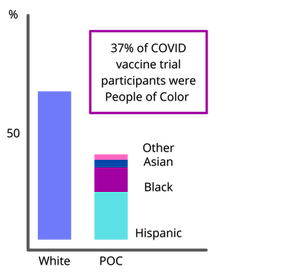
Over 200,000 Americans safely took part in trials, and more than twice that many people were tested world-wide.
After a history of failing to adequately include all racial and ethnic minorities in trials, the COVID vaccines are the first to prioritize and achieve samples that fully reflected the American public.
Studies found the COVID vaccine works as well in racial and ethnic minorities as in white Americans. No Person of Color who received the vaccine later contracted COVID.
Most trial participants had soreness in their arm like with the flu vaccine and other vaccines. Some had soreness and body aches that lasted longer than a day.
People of different races and ethnicities were no more or less likely to experience side-effects of the vaccine. Older Americans were slightly less likely to report side-effects.
Full FDA Approval
After months of additional, rigorous review, Pfizer-BioTech’s COVID vaccine received full FDA approval on August 23, 2021. Pfizer COVID Vaccine approval was based on safety and effectiveness data from a randomized, controlled, blinded ongoing clinical trial in approximately 18,000 individuals who received the vaccine and approximately 18,000 who received a placebo.
Moderna is expected to be approved soon.
Vaccine Distribution
Who Makes It?
- After extensive testing, Pfizer-BioTech’s vaccine has received full FDA approval.
- Moderna and Johnson & Johnson have received emergency use FDA approval. They are expected to receive full approval in the coming months.
- Moderna, Johnson & Johnson and Pfizer are all U.S. companies.
Who Takes It?
- Over 200 million Americans have taken the vaccine
- 5.35 billion doses have been given worldwide.
- 1 million Americans are getting the COVID vaccine each day
- Any American 12+ is eligible for a COVID vaccine
Who Created the COVID Vaccine?
To learn more about the minds and science behind the vaccine, visit the bio of our COVID vaccine expert.
Scientific Lead,
NIH COVID Vaccine Program
Sign Up
To request more information about COVID-19 and FLU vaccines, please fill out this form.
By submitting this form, you agree to receive text messages for purposes related to vaccine information only. Message and data rates may apply.